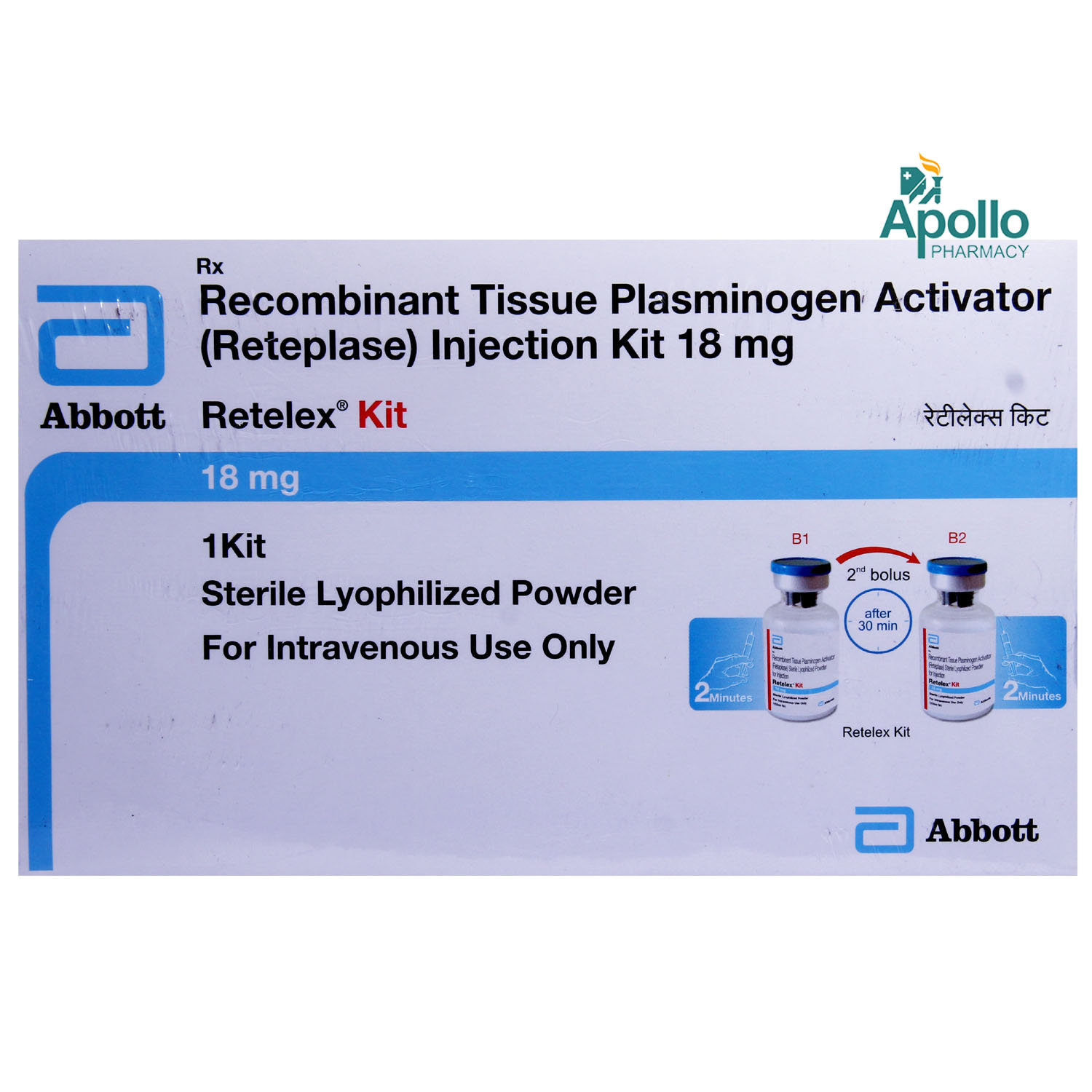
Reteplase
About Reteplase
Reteplase is a tissue plasminogen activator indicated for the treatment of acute ST-elevation myocardial infarction (STEMI) to lower the risk of heart failure and death. STEMI is a very serious type of heart attack that occurs due to blockage of the heart’s major arteries.
Reteplase contains Reteplase, which works by dissolving the blood clot that is blocking the heart blood vessel.
In some cases, Reteplase may cause side effects such as bleeding, bruising at the injection site, nausea or vomiting. You are advised to talk to your doctor if any of the side effects persist or worsen.
Consult your doctor if you are pregnant or breastfeeding. The efficacy and safety of Reteplase in children have not been established. Inform your doctor about all the medicines you are taking and your health condition to rule out any unpleasant side effects/interactions.
Uses of Reteplase
Medicinal Benefits
Reteplase is a tissue plasminogen activator indicated for the treatment of acute ST-elevation myocardial infarction (STEMI) to reduce the risk of heart failure and death. Reteplase contains Reteplase that works by dissolving the blood clot that is blocking the heart blood vessel. It may also be indicated in the treatment of a recent left bundle branch block (a delay/blockage of electrical impulses to the left side of the heart).
Directions for Use
- Follow your doctor's instructions on the dosage and timing of this medication to ensure safe and effective use.
- Reteplase will be administered by the doctor or healthcare professional.
- Do not self-administer.
Storage
Side Effects of Reteplase
- Bleeding
- Bruising at the injection site
- Vomiting
- Nausea
Medicines Containing this Salt
View AllDrug Warnings
Reteplase should not be used if you are allergic to any of the components in Reteplase, in patients with active internal bleeding, recent stroke, recent intracranial or intraspinal surgery or serious head trauma, intracranial neoplasm (brain tumour), arteriovenous malformation, or aneurysm (ballooning and weakened area in an artery), known bleeding diathesis (tendency to bleed or bruise easily), or severe uncontrolled hypertension. Reteplase may cause bleeding, hypersensitivity reactions, or cholesterol embolism. Inform your doctor if you have/had recent major surgery, cerebrovascular disease, recent gastrointestinal/genitourinary bleeding, hypertension, subacute bacterial endocarditis, severe hepatic or renal disease, if you are over 75 years or if you are taking any oral anticoagulants. Consult your doctor if you are pregnant or breastfeeding. The safety and effectiveness of Reteplase in children have not been established.
Drug Interactions
Drug-Drug Interactions: Inform your doctor if you are taking anti-coagulants (heparin, enoxaparin, warfarin, phenindione, aspirin, dipyridamole, ticlopidine, clopidogrel, vitamin K antagonists).
Drug-Food Interactions: No interactions found/established.
Drug-Disease Interactions: Inform your doctor if you have/had cancer, stroke, high blood pressure, active ulcer in the stomach or intestine, portal hypertension, inflammation of the pancreas, bleeding behind or in the eye, or severe liver or kidney problems.
Drug-Drug Interactions Checker List:
Safety Advice
Alcohol
consult your doctorLimited information is available. Please consult your doctor.
Pregnancy
cautionPlease consult your doctor if you are pregnant. Your doctor will prescribe only if the potential benefits outweigh the potential risk to the foetus.
Breast Feeding
cautionPlease consult your doctor if you are breastfeeding. Caution should be exercised whilst administering Reteplase to a nursing mother.
Driving
consult your doctorLimited information is available. Please consult your doctor.
Liver
cautionThe potential risks with the therapy of Reteplase may be increased in patients with severe liver dysfunction and should be weighed against the anticipated benefits. Please consult your doctor.
Kidney
cautionThe potential risks of Reteplase therapy may be increased in patients with severe renal dysfunction and should be weighed against the anticipated benefits. Please consult your doctor.
Children
cautionThe efficacy and safety of Reteplase in children have not been established.
Habit Forming
Diet & Lifestyle Advise
- Include vegetables, fruits and whole grains in your diet.
- Stop smoking.
- Be physically active every day.
- Aim for a healthy weight.
- Limit the consumption of alcohol.
- Manage stress with meditation.
Patients Concern
Disease/Condition Glossary
ST-elevation myocardial infarction (STEMI): It is a very serious type of heart attack that occurs due to blockage of the heart’s major arteries. Signs and symptoms include dizziness or light-headedness, chest pain or discomfort, shortness of breath, nausea or vomiting, palpitations, anxiety, or sweatiness unexplained by ambient temperature.
FAQs
Reteplase is used to treat ST-elevation myocardial infarction (STEMI).
Reteplase works by dissolving the blood clot that is blocking the heart's blood vessels.
Reteplase may increase the risk of bleeding. The sites of bleeding could be internal or external. Talk to your doctor if you have any concerns.
Heparin and Reteplase are incompatible. Reteplase should not be administered through an intravenous line containing heparin. Please consult your doctor if you have any concerns.
Reteplase will be administered by a healthcare professional in a clinical setting. Do not attempt to administer it yourself. A trained healthcare professional will ensure safe and proper administration.
Yes, Reteplase is safe when used under the doctor’s supervision. It is used in the treatment of ST-elevation myocardial infarction (STEMI). However, it may cause some side effects like bleeding, bruising at the injection site, nausea, or vomiting in some cases. You are advised to talk to your doctor if any of the side effects persist or worsen.
Reteplase may interact with anti-coagulants like heparin, enoxaparin, warfarin, phenindione, aspirin, dipyridamole, ticlopidine, clopidogrel, and vitamin K antagonists. So, it’s best to consult your doctor before taking anti-coagulants or any other medications along with Reteplase.
Reteplase helps prevent blood clots from forming in your legs, lungs, heart, and brain. You may not feel any immediate effects after taking the medicine, but that doesn't mean it's not working. Continue taking the medicine exactly as prescribed by your doctor, even if you don't notice any difference.
The common side effects of Reteplase may include bleeding, bruising at the injection site, nausea, or vomiting. You are advised to talk to your doctor if any of the side effects persist or worsen.