Living with type 2 diabetes often involves ongoing attention to blood sugar levels, medications, and lifestyle choices. As treatment options continue to evolve, newer therapies are offering additional ways to improve control and reduce long-term health risks.
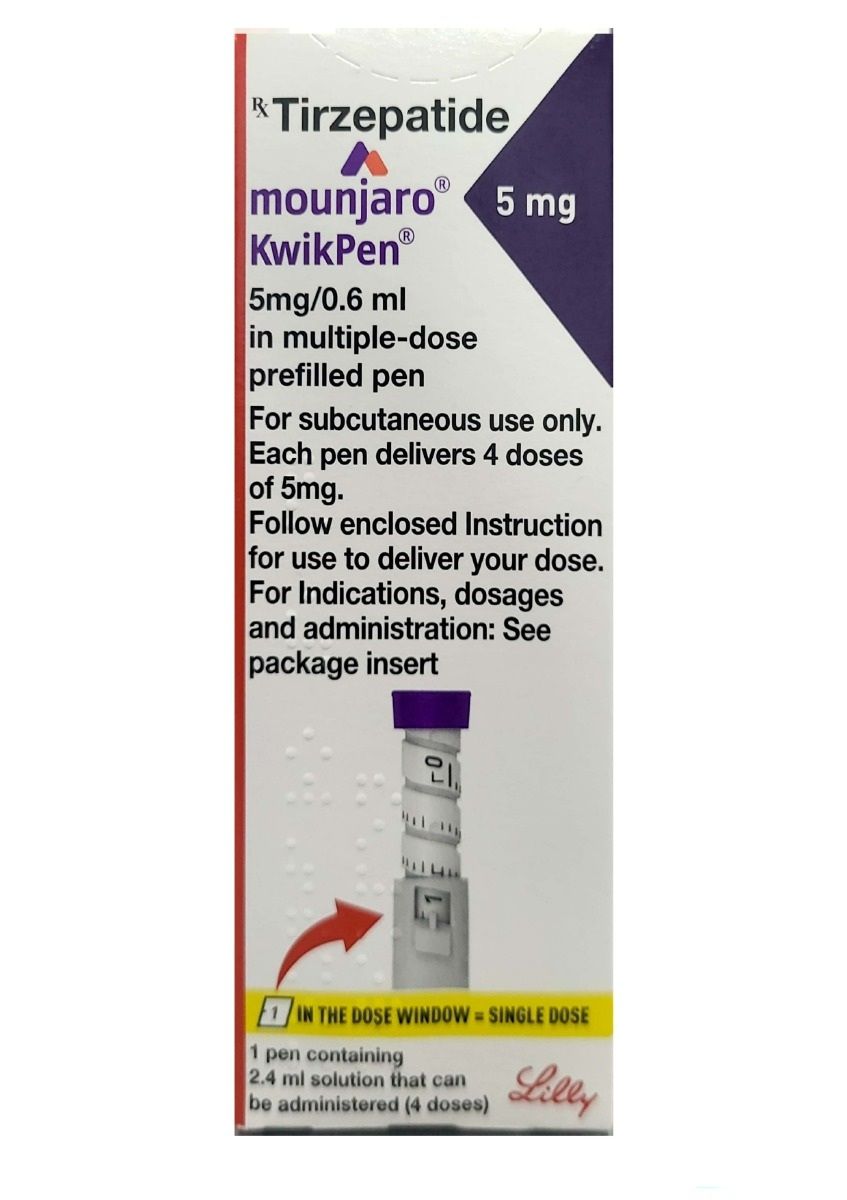
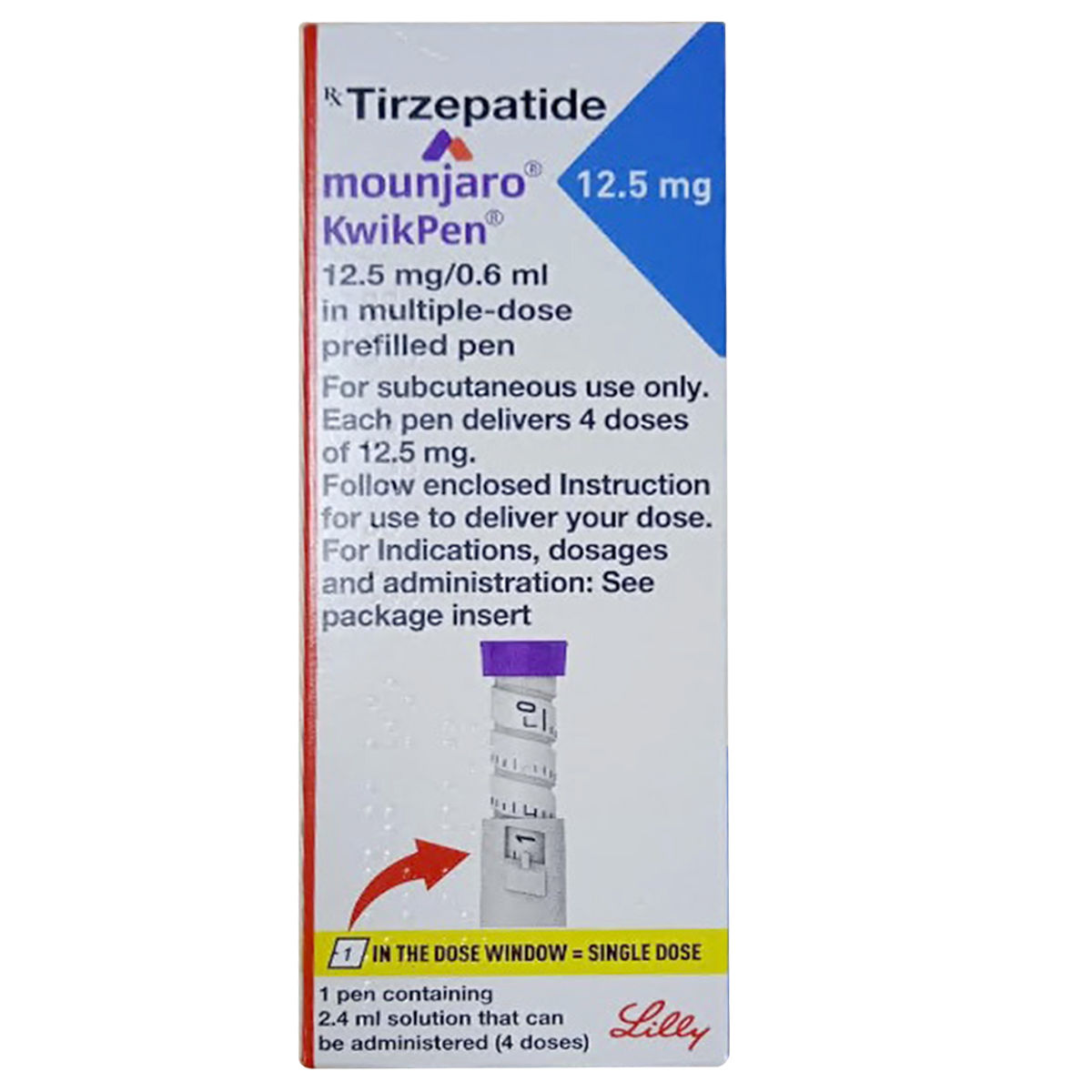
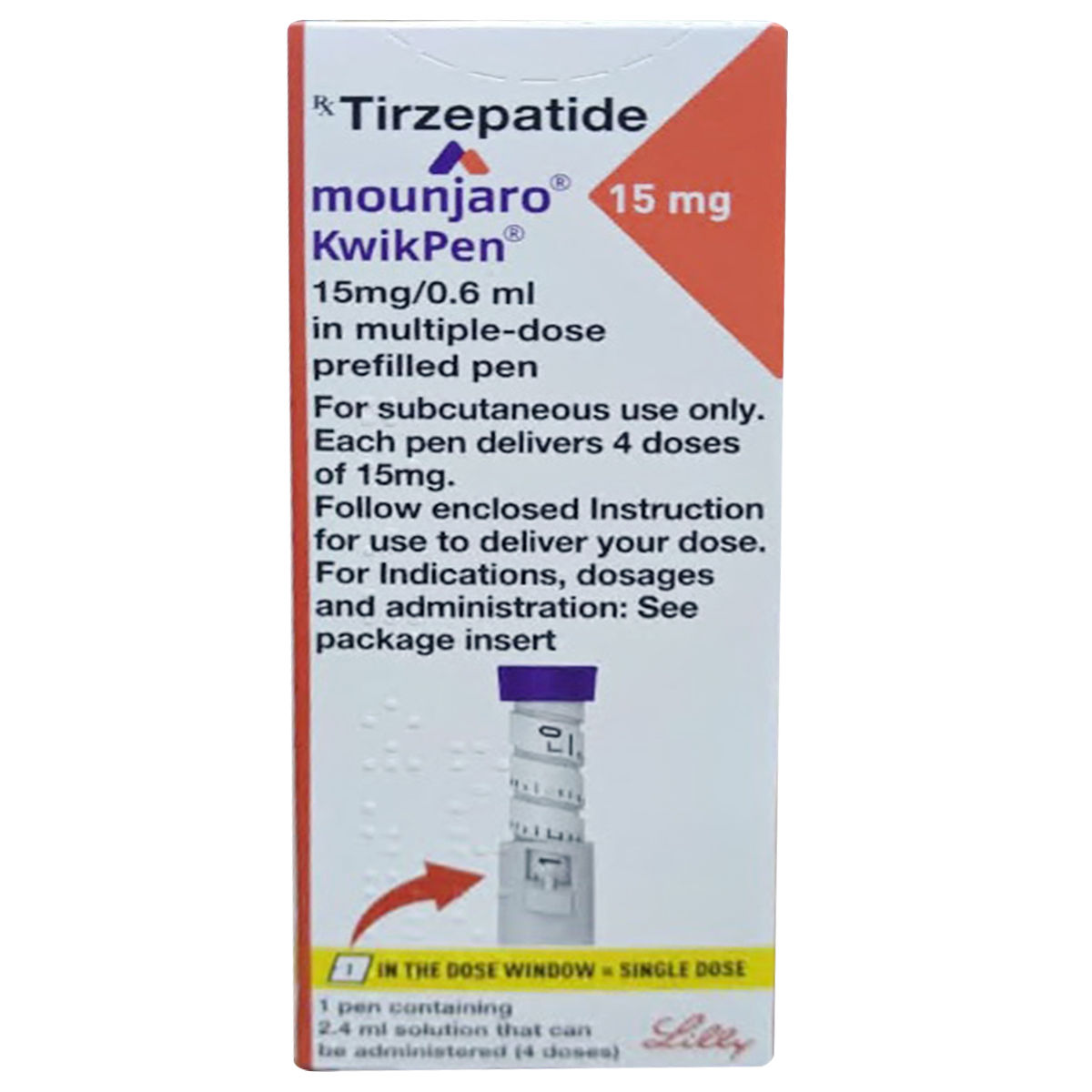
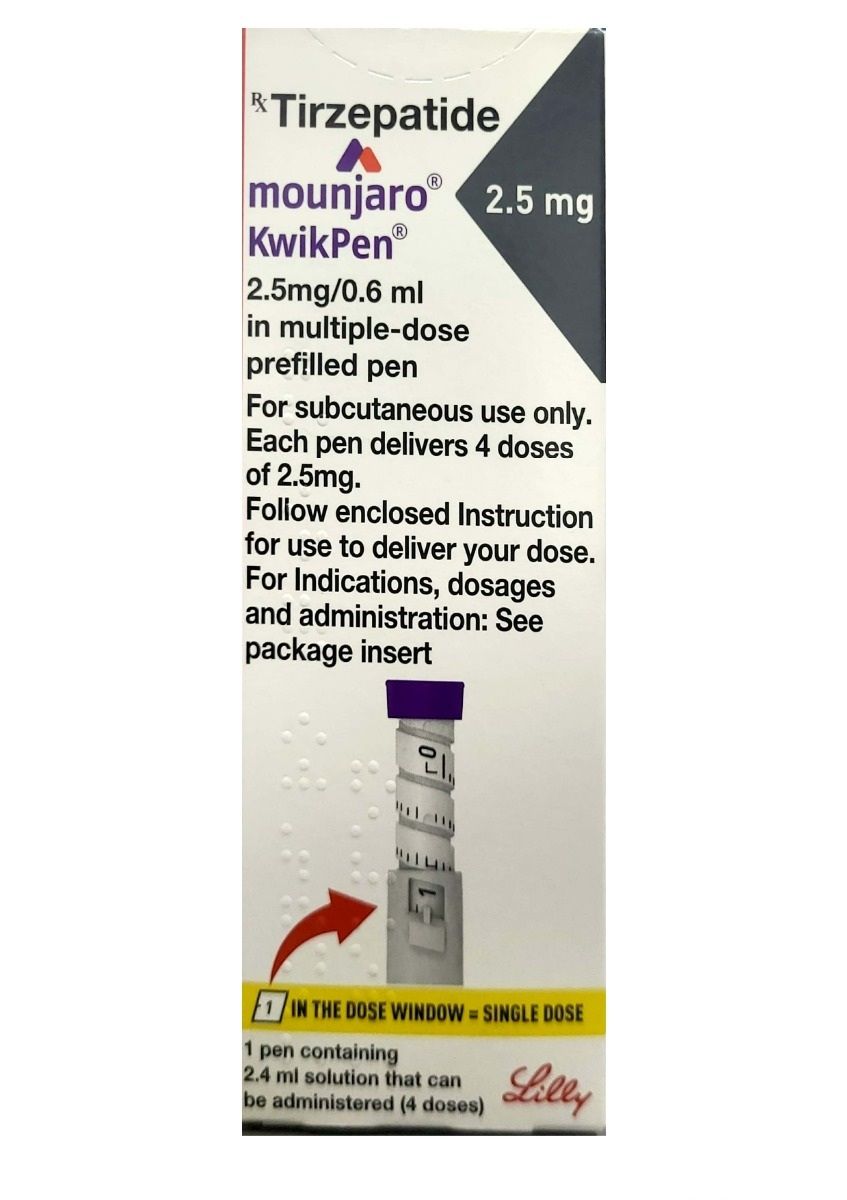
Mounjaro (tirzepatide) is a prescription medication approved for the treatment of type 2 diabetes. It is the first therapy in its class to activate two gut hormone pathways, glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors, providing a different approach to blood sugar management compared with earlier treatments that target a single pathway.
Clinical studies have shown that tirzepatide can significantly improve glycaemic control while also supporting weight reduction and broader metabolic benefits in appropriate patients. These effects have made Mounjaro an important option for people who need additional support beyond lifestyle measures and standard diabetes medications.
This guide explains how Mounjaro works, how it is used, its potential benefits and risks, and key safety considerations, helping patients have informed discussions with their healthcare provider.